Digestion is a physiological process by which ingested food is broken down into simple, small and absorbable molecules with the help of enzymes. These enzymes are secreted from the pharynx, stomach, pyloric Caeca, intestinal mucosa and pancreas into the esophageal cavity.
Digestive Juice and Enzyme
Digestive juices and enzymes are part of the stomach and pancreatic, bile and intestinal secretions. With the exception of some fish without a stomach, most fish have gastric juice. The intestines of fish without stomach do not produce hydrochloric acid (HCl) or pepsin. The following table lists the digestive juices and enzymes secreted by teleost fish:
Table: Digestive juices and enzymes secreted by teleost fish
|
Site/type |
Juice/enzyme |
Function |
|---|---|---|
| Stomach | ||
|
Gastric secretion |
HCl |
Decreases the pH value of the stomach and activates pepsinogen. |
| Gastric gland |
Zymogen Pepsinogen, HCl Pepsin |
Proteolytic enzymes: Causes the division of aromatic and acidic amino-group (-NH2) peptide chains, breaking down most proteins. |
|
Amylase |
Acts on carbohydrates | |
|
Lypase |
Acts on fat | |
|
Esterase |
Acts on Easter | |
|
Chitenase |
Acts on chitin | |
| Pancreas |
Enzymes |
Enzymes are stored as zymogens. Protease originates from the intestine and converts trypsinogen to trypsin which activates others. |
|
HCO3 |
It neutrals HCl entering the intestine and prepares the intestine for alkaline digestion. | |
|
Proteases (trypsin, chymotrypsin, carboxy peptidase and elastase |
These enzymes react moderately at pH 6.0. | |
|
Trypsin |
Divides the peptide chain of the carboxy group of lysine or arginine. | |
|
Chymotrypsin |
Acts on the carboxylic peptide. | |
|
Elastase |
It acts on elastin peptide bonds. | |
|
Carboxypeptidase |
It hydrolyzes the marginal peptide bonds of substrate. | |
|
Amylase |
It digests carbohydrates without pH. | |
|
Chitinase(+NAGase) |
It converts chitin to dimer and trimmer of N-acetyl-D-glucosamine (NAG) which is then broken down again by NAGase. | |
|
Lipase |
It hydrolyzes triglycerides, fats, phospholipids and wax esters. | |
| Bile (Secreted from the liver) |
Bile salts, organic anions, cholesterol, phospholipids, inorganic ions |
It alkalizes the intestinal medium and emulsifies lipids. Most bile salts are reabsorbed through the intestines and reach the liver through enterohepatic circulation. |
| Intestinal enzymes (secreted from the brush border region of the epithelium, but may originate from the pancreas partially) |
Aminopeptidase (alkaline and non-alkaline) |
It analyzes nucleosides. |
|
Polyneucleotidase |
It analyzes nucleic acid. | |
|
Lecithinase |
It breaks down phospho lipids into glycerol and fatty acids. | |
|
Different types of carbohydate digesting enzymes |
It breaks down carbohydrates |
A digestive enzyme called hydrolase acts as a catalyst in the hydrolysis reaction. This enzyme consists of water soluble proteins. Hydrolase enzymes are divided into proteases, lipases, esterase and carbohydrase based on physiological functions. This enzyme can be produced in other tissues besides the main region, such as amylase is also produced in the liver. It has also been found that enzyme-rich animals supply a significant portion of fish food which may increase the endogenous activity of the enzyme. This condition is especially seen in immature fish.
Most of the food digestion in fish is extracellular which is done in the cavity of the alimentary canal. Also a significant amount of digestion depends on the inactivation of cell membranes by enzymes. This process is mostly seen in the middle and end stages of digestion and is considered as a link in the assimilation strategy.
Digestion of Protein in Fishes
Proteins are larger molecules made up of chains of amino acids. Although there are more than 200 amino acids in nature, 20 of which are very common. Of all these amino acids, 10 are essential that fish cannot synthesize. These are methionine, arginine, threonine, tryptophan, histidine, isoleucine, lysine, leucine, valine and phenyl alanine. Protein contains 50% carbon, 18% nitrogen, 21.5% oxygen and 6.5% hydrogen.
The secretion of hydrochloric acid (HCl) and pepsin initiates the digestion of proteins in the stomach. Pancreatic enzymes such as trypsin in the duodenum and upper jejunum that break down most undigested proteins into the smallest unit of a short chain of amino acids, the tiny peptide with 2-6 amino acids. Some of these tiny peptides are directly absorbed. As there is no enzyme that digests protein in the mouth, there is no digestion of protein here. Only physical changes in the protein are achieved here.
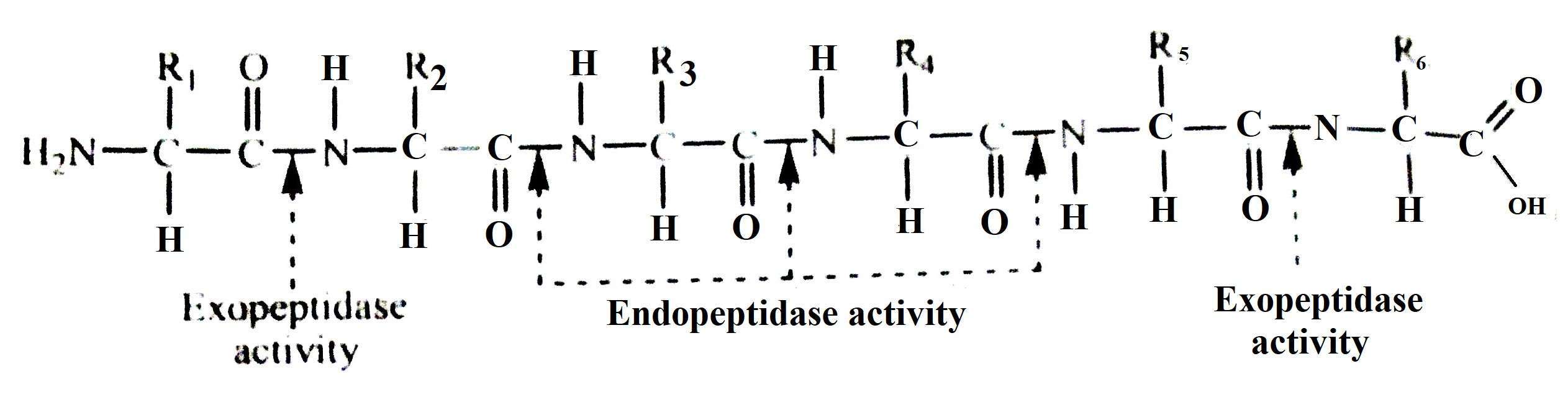
Proteolytic enzymes are produced from an inactive substance called zymogen. Zymogens are processed in the intestinal cavity through hydrolysis of acid or proteolytic reactions. It protects tissues from self-digestion. Protease breaks down the peptide chain of proteins. Different types of enzymes are able to act on the end of the peptide bond of the protein (exopeptidase) or in (endopeptidase). The enzyme endopeptidase and exopeptidase act on different parts of the peptide chain during hydrolysis of a simple protein which is shown in the following reaction:
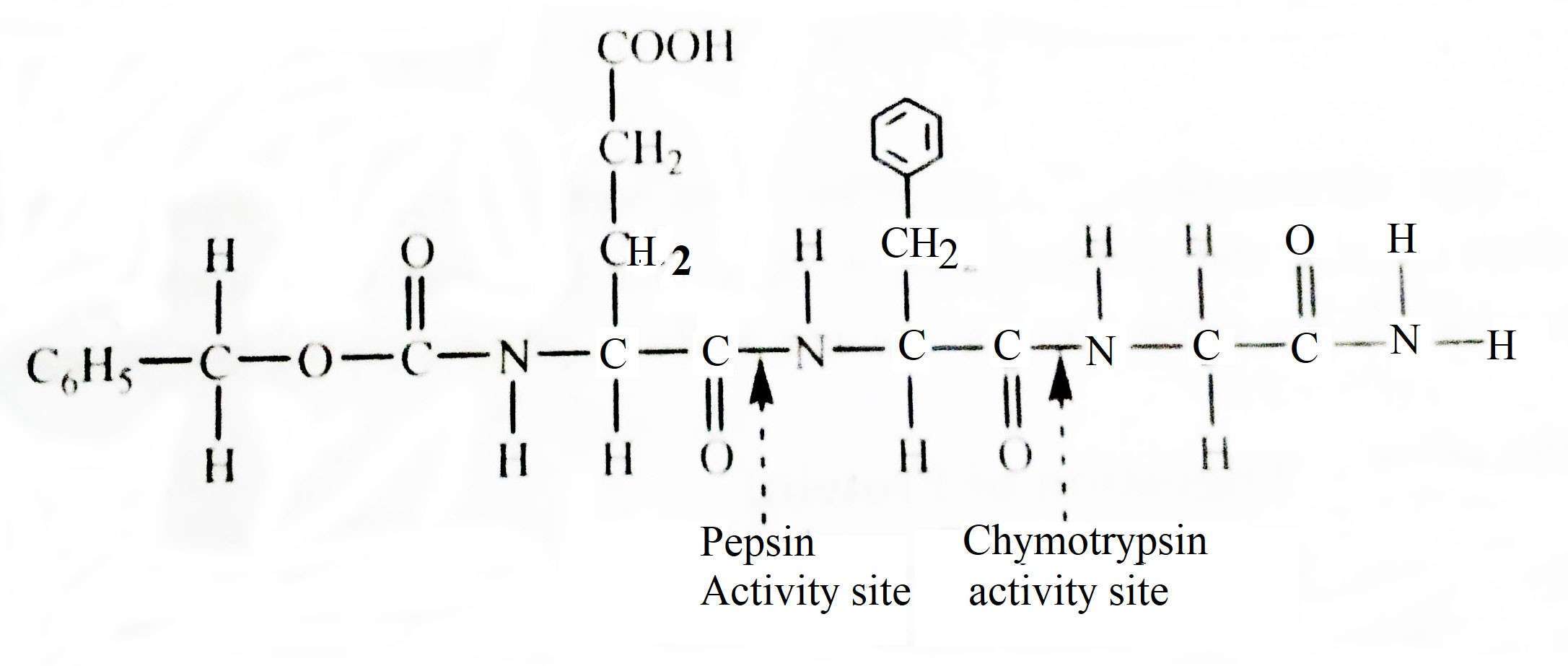
Endopeptides are very specific in function and they only act on a specific point of the protein molecule. The action of a particular endopeptidase is determined based on the nature of the chemical group on both sides of the corresponding bond. A bond that has already been semi-analyzed by an endopeptidase may be completely resistant to hydrolysis for other enzymes. The breakdown of proteins and the chemical nature of the products produced are determined by the existing endopeptidases. The following example shows a hydrolysis of a synthetic protein called benzyl-oxycarbonyl-L-gutamyl-L-tyrosilglycin amine.
In the above hydrolysis process, it is seen that pepsin analyzes the amino side bonds of aromatic radicals and chymotrypsin carboxyl side bonds.
Trypsin, on the other hand, acts on the peptide bond between arginine and lysine. Endopeptidase enzymes called chymotrypsin, trypsin and pepsin play a more important role in protein digestion. These break down most of the proteins ingested into food and turn them into polypeptides.
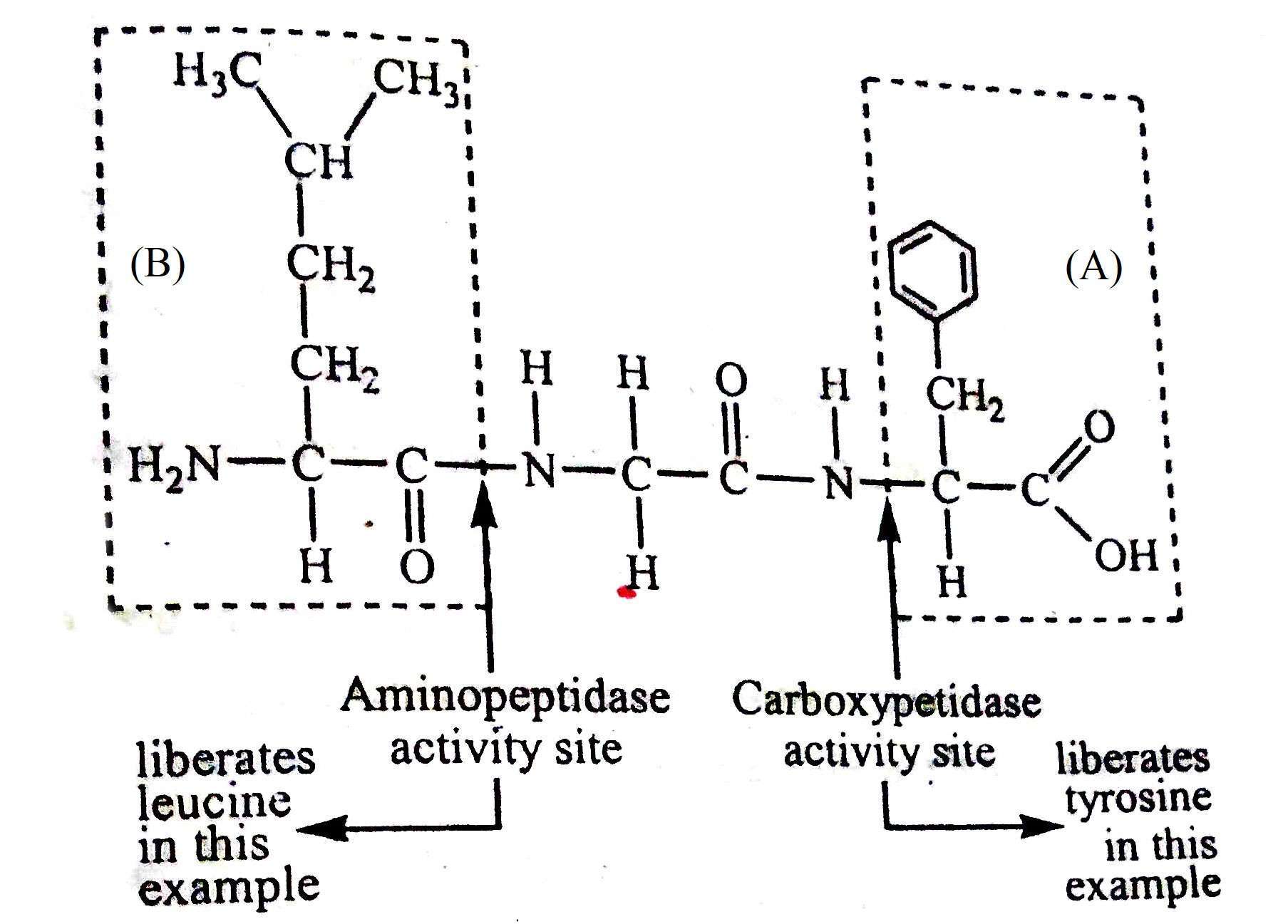
There are three types of exopeptidase enzymes, namely, carboxypeptides, aminopeptidase and dipeptidase. Each of these acts on a specific type of substrate or substrate group. Like endopeptidase, this specificity is determined by the nature of the groups on each side of the peptide bond (which will be hydrolyzed). In the case where carboxyl radicals are free, carboxypeptides remove marginal amino acids as shown in the diagram below (a). Aminopeptides, on the other hand, act on the other end of the polypeptide chain to remove endogenous amino acids with free amino groups, as shown in Figure (b).
Some fish are able to absorb dipeptides, even larger polypeptides, which break down intracellularly into distinct amino acids.
Both cholinergic and adrenergic nerves exist in the stomach which stimulates the secretion of gastric juice. Pepsin is a protease enzyme that can break down proteins. The intestinal mucosa of carnivorous fish secretes hydrochloric acid (HCl) which helps in creating low pH values. Most of the digestion of protein in the stomach is completed by the action of pepsin. Inactive pepsinogen is converted to active pepsin under the influence of hydrochloric acid (HCl) in the presence (in some cases absent) of food in the stomach. Pepsin is more active in acidic solutions. In herbivores (tilapia), it is thought that the presence of acidic medium also helps to break down plant cell walls. Partial crushing of food taken through the pharyngeal teeth facilitates hydrolyzes of the cell wall by HCl so that proteolytic enzymes are more able to act on plant cells.
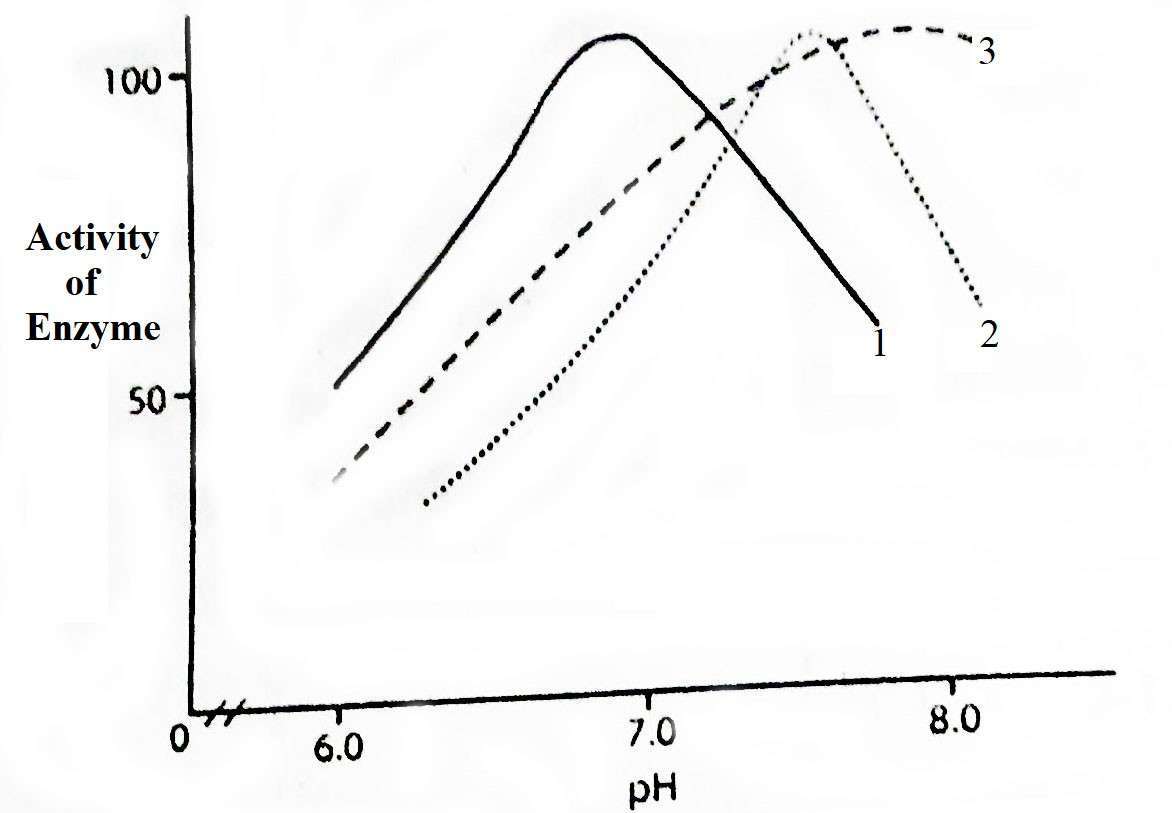
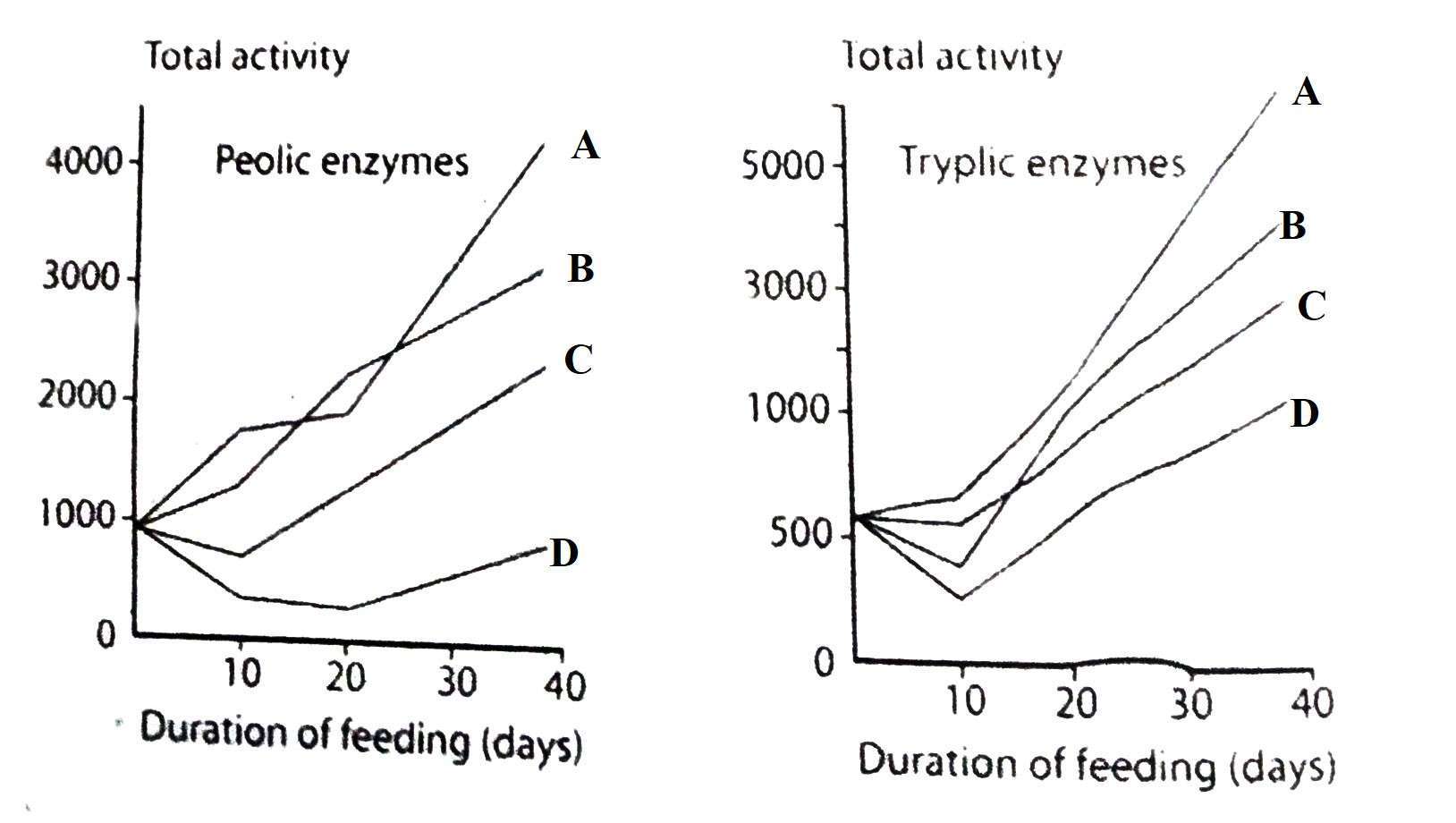
Figure: Relation of pH with the activity of intestinal enzymes of rainbow trout. 1, amylase; 2, estrogen; 3, Protease (Steffens 1989)
Pepsin plays the role of alkaline protease in stomachless fish. These proteases are more active in alkaline environments. The stomach has evolved periodically in all species of fish. Incomplete digestion of the protein is performed until it is fully developed. Similarly, low activity of alkaline proteases is observed in fish that have no stomach. In general, the digestion of proteins in the early immature stages of fish is more dependent on alkaline tryptic enzymes than on acidic peptic enzymes. Pancreatic extract of some elasmobranch contains trypsin.
In carp, it has been shown that the action of some of these enzymes is related to food. As the proportion of fish meal in the diet decreases, the activity of protease decreases. On the other hand, increasing the amount of starch in the diet increases the activity of amylase.
The activity of proteases in the hepatic saeca of rainbow trout depends on the temperature and quality of the food. In the case of low protein diets, no significant effect of temperature has been observed. Protein activation also changes with the change in food content in the rainbow trout.
Figure: Effect of food ingredients on the level of overall activation of proteolytic enzymes in baby rainbow trout; (a) 80% fish meal, 0% a- cellulose food; (c) 40% fish meal, 40% -a- cellulose food; (d) 20% fish meal, 60% -a- cellulose food(Kawai and Ikeda 1973).
Table: Activation of intestinal proteolytic enzymes in common carp fry
| Age(day) |
Time* |
Weight(mg) |
Protease activation (microgram /gram) |
|---|---|---|---|
|
8 |
5 |
10.5 |
42 |
|
17 |
14 |
57 |
219 |
|
22 |
19 |
114 |
266 |
Time* From the start of protein intake. Source: Steffens (1989)
Digestion of Lipid/Fat in Fishes
Fat is a type of organic material. It is made up of numerous carbon molecules in different types of chains. Fats are insoluble in water but soluble in various organic solvents such as chloroform, ether and benzene. In animals, it plays an important role as a high calorie storage molecule or cell membrane component. Fats can be divided into five main categories, namely, fatty acids, triglycerides, phospholipids, sterols and sphingolipids. These fats contain a variety of vitamins and essential fatty acids. An enzyme called lipase participates in the digestion of lipids. Such enzymes are found in the pancreas and mucosa of fish. Lipase breaks down fats into fatty acids and glycerol.
The liver plays an important role in fat digestion. Bile is produced in the liver and stored in the gallbladder and is blocked when food reaches the intestines. Fats contain more active Gallic acid (also known as 3,4,5-trihydroxybenzoic acid) and it emulsifies fats to break down larger fat particles into smaller ones. Moreover, it increases the reaction level and creates a conducive environment for fat-soluble enzymes. All types of fat-digesting enzymes are classified as lipolytic enzymes or lipase. Evidence of lipase activation has been found in the extract of the pancreas, pyloric caeca, and upper intestine. However, it is not essential for all fish to have these enzymes in these three places. Activation of lipase in the stomach was not known. However, the intestinal mucosa layer is considered to be the main area of lipase. Lipase shows lower substrate specificity in hydrolysis than in carbohydrates and proteins. Any organic Easter acts as a catalyst in most hydrolysis. The progressive division of fats through different types of interstitial phases is accomplished only by lipase enzymes, and here different types of enzymes, such as proteolysis, are not specific.
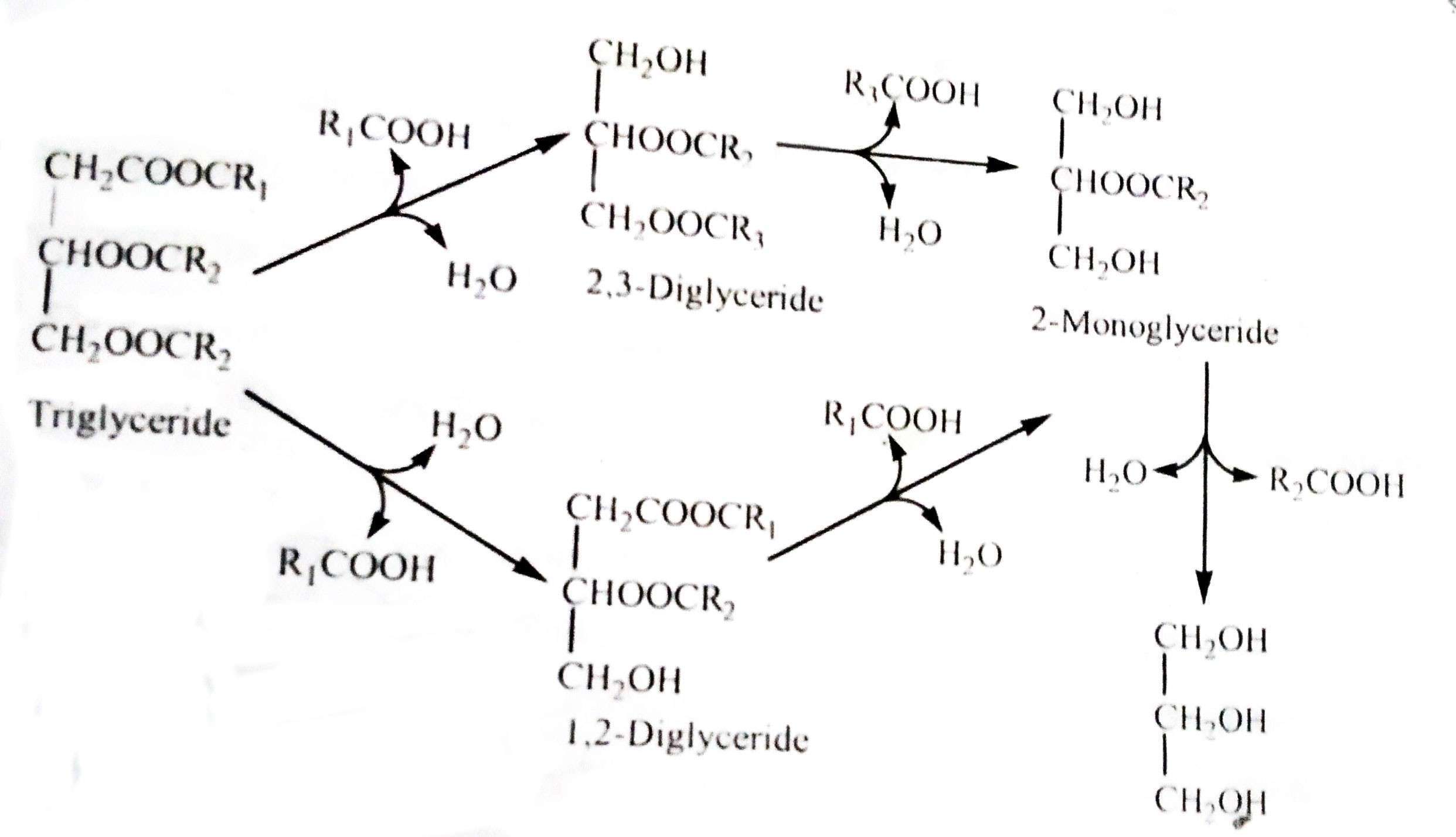
Fats are commonly used by animals as esters of organic acids and superior alcohols (usually glycerol, a trihydric alcohol). In the final stage, the fat is dissolved to form a triglyceride molecule consisting of three molecules of fatty acids and one molecule of glycerin. The process of fat analysis is shown in the following reaction:
All types of fat digesting enzymes work through alkaline medium. Slight variation in the moderate pH value was observed in different groups. The lipase secreted from the intestinal mucosa is able to function well in the pH range of 7-7.5. However, intestinal esterase pH values are more active between 8-9.
Digestion of Carbohydrate in Fishes
Carbohydrate is known as sugar or saccharide which is an essential element of all organisms. It plays an immediate role in storing metabolic energy as a molecule and helps transfer energy through the organism as a structural element. The basic unit of carbohydrate is known as monosaccharide. In the biological system, monosaccharides are usually produced by glucogenesis or photosynthesis. Monosaccharides are found as the main constituents of nucleic acids or are chained to form polymer compounds. Such polymer compounds can be divided into two distinct groups, namely oligosaccharides and polysaccharides.
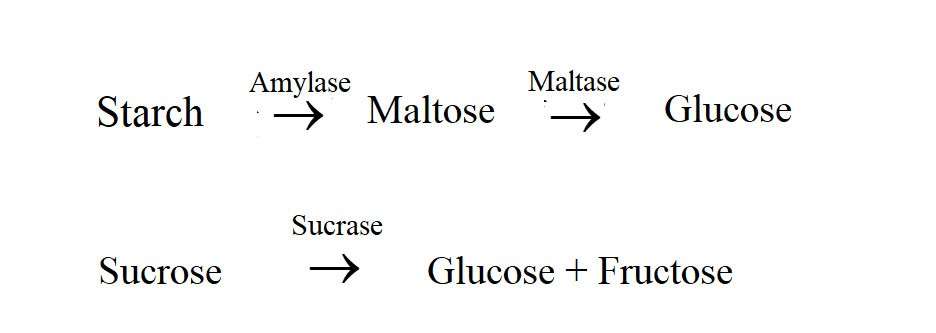
The intestines of fish contain a significant number of different types of carbohydrate-digesting enzymes that have specific functions. The enzymes in the intestines of fish that participate in the digestion of sugars are called carbohydrates. Hydrolase enzymes include amylase, lactase, saccharase / sucrase and cellulase. Carbohydrates are a type of high (200-400 C) temperature tolerant enzyme that works well at pH 6-8. Like carbohydrase lipase, it is found in pancreatic juice, stomach, intestines and bile. The pancreas is the main producer of carbohydrase in most species. Amylase is a very important enzyme that acts on starch and converts it into maltose. It was later converted to glucose by Maltese. In carnivorous fish, amylase is secreted from the pancreas, but herbivorous fish secrete enzymes throughout the stomach and intestines, including the pancreas. Like tilapia, herbivores have amylase in their entire digestive tract. These fish contain an enzyme called sucrase, which reacts with sucrose to produce glucose and fructose. The following are some of the digestive reactions that are shown sequentially:
Blood glucose is converted to glycogen with the help of insulin and accumulates in the muscles. Although the details are not known, it is thought that excess glucose from the digestive tract enters the bloodstream and is converted to glycogen in the liver.
There are a number of specific carbohydrase enzymes that convert starch or glycogen into oligosaccharides or maltose into mono and polysaccharides with the help of a-amylase by hydrolysis of di and oligosaccharides.
Depending on the structure of the substrate, these molecules are broken down by glucosidase, galactosidase and fructosidase into simpler components. Carbohydrate activation responds to dietary sugar levels. The amylase activity in rainbow trout increases with food intake.
On the other hand, feeding tilapia (Oreochromis mossumbicus) rich in starch increases amylase activity. In the case of Oreochromis niloticus, low sugar levels respond to high-starch foods, but an increase in α-glucosidase and β-galactosidase responds to an increase in lactose levels in the diet. Common carp show the opposite effect of rainbow trout to the levels of starch in the diet. In general, the activity of carbohydrase, especially amylase, varies from species to species. The activity of other carbohydrase such as chitinase varies between species.
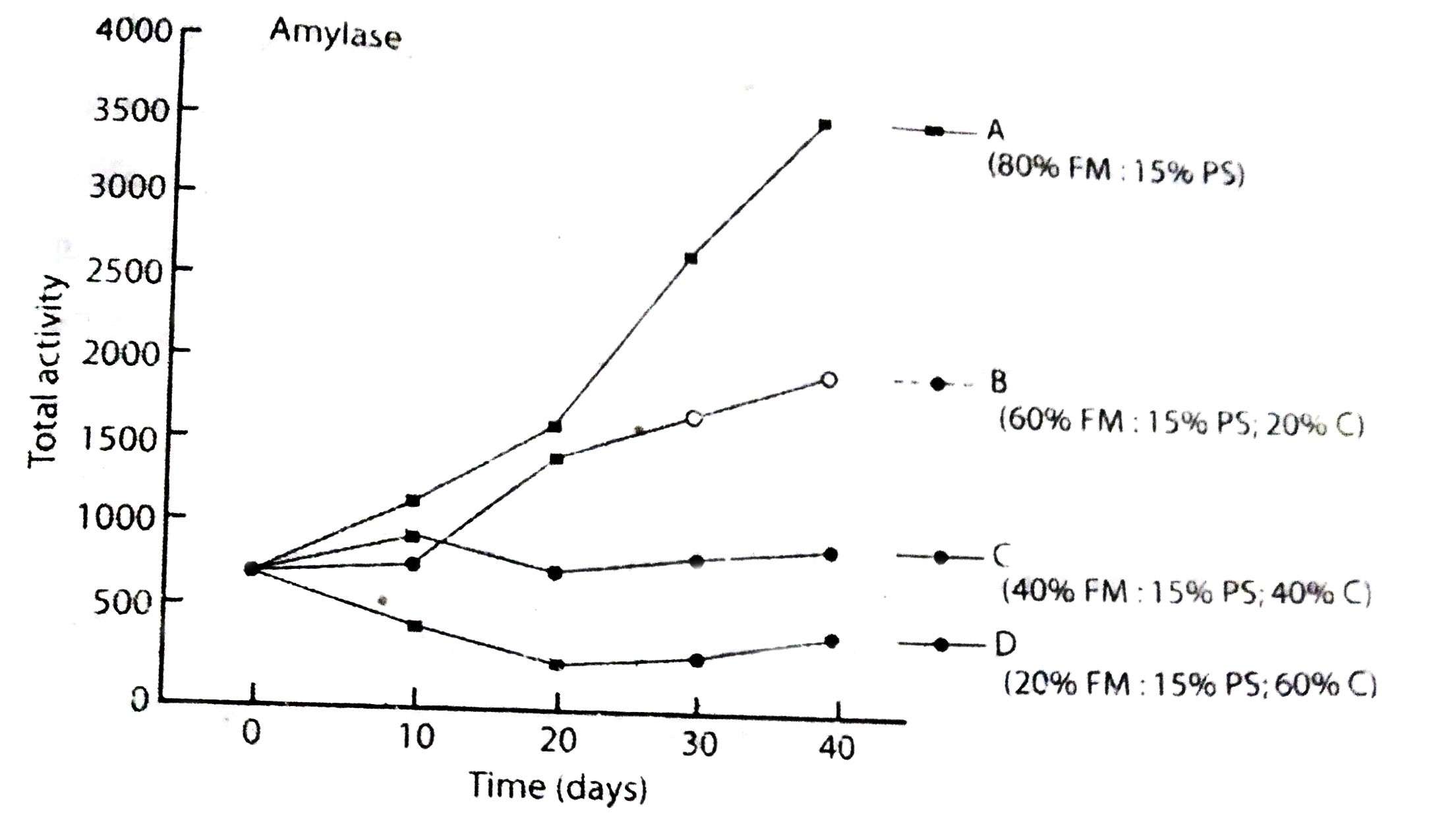
Fig.: Activation of amylase in various foods in rainbow trout: fish meal (FM); potato starch (PS); Cellulose (C) (Kawai and Ikeda 1973); As the level of fish meal in the diet increases, so does the level of protein.
Table: Relative activity of amylase, α-glucoccidase and β-gactocydase in the digestive tract of different species (maximum = 100)
| Species | Amylase | α-glucosidase | β-glucosidase | ||||
| Intestine | Stomach | Pyloric Caeca | Intestine | Stomach | Intestine | Stomach | |
|
Carassius carassius |
100 | 34 | |||||
|
Ctenopharyngodon idellus |
84 | 100 | 61 | ||||
|
Oreochromis niloticus |
44 |
31 | 59 | ||||
|
Cyprinus carpio |
35 | 8 | |||||
|
Hypophthalmicthys molitrix |
31 | 100 | |||||
|
Salmo gairdneri |
8 |
<1 |
16 | 2 | <1 | ||
|
Anguilla japonica |
1 |
<1 |
15 | 20 | 11 | ||
|
Seriola quinqueradiata |
1 |
<1 | 22 | 6 |
In general, the activity of carbohydrase and protease depends on the diet of the fish. Carnivorous fish naturally have higher levels of enzymes that digest protein and other fish have lower levels of enzymes. Differentiation of proteolytic enzymes between species is less important than amylase.
Microbial Digestion in Fishes
In some animals, Microbial digestion plays an important role in digestion of cellulose and protein synthesis. Endogenous bacteria produce cellulose. Not much research has been done on this. However, the research of Stickney and Shumway (1974) is noteworthy in this context. They conducted research on 62 species belonging to 35 genera and observed some cellulose activity in 17 species. In this case, they noticed that the presence of enzymes does not depend on intestinal morphology or eating habits.
Evidence suggests that cellulose activity in fish is caused by microbial flora. Bacteria are usually found in the intestines of herbovores and detritus feeders. Significant amounts of proteolytic and amylolytic enzymes can be observed in these fish. Activation of chitinase and lecithinase can also be observed in microorganisms. In this context, Prejs and Blaszczyk (1977) observed in their study that there is a positive relationship between the amount of plant food in the stomach of cyprinids and the activation of cellulose.
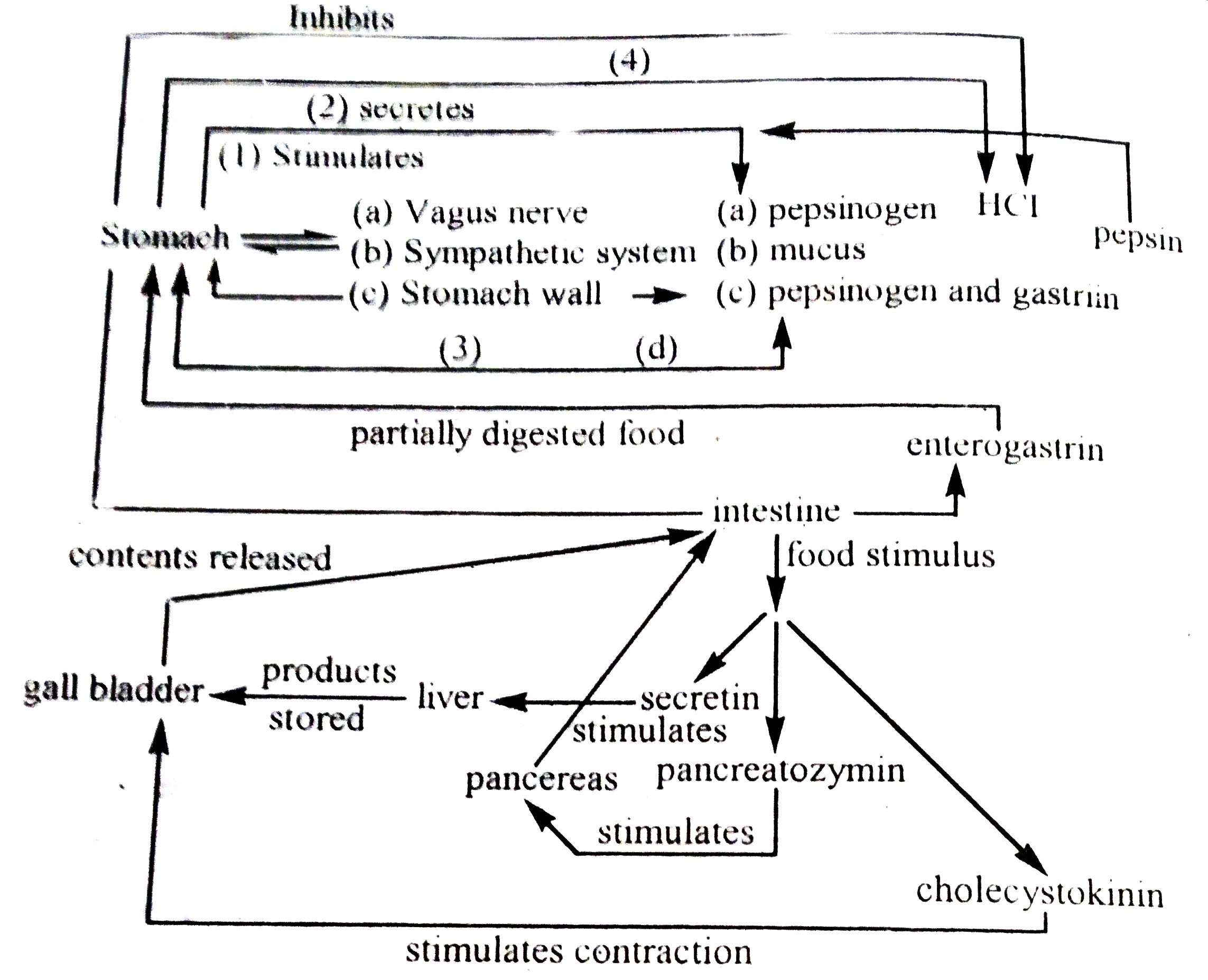
Hormone in Digestion of Fishes
Four types of hormones are present in the mucosa of the gastrointestinal tract. These are secretin, cholecystokinin, gastrin and gastric inhibitory peptide. Each of these is generated from gastrointestinal endocrine cells and is transported throughout the body through the bloodstream to the plasma membrane receptors of specific cells. The presence of gastrin and cholecystokinin in teleost has been proven. These hormones are secreted from scattered intestinal endocrine cells. Cholecystokinin affects oxytocin cells and inhibits intestinal secretion again.
Somatostatin is present in the stomach and pancreas of fish. It is also called paracrine object. It differs from hormones in that it does not travel through the blood but it spreads directly to specific localized cells. It inhibits gastrointestinal and pancreatic islet cells with other endocrine features.
The presence of vasoactive intestinal peptides and pancreatic peptides in the digestive tract of Puntius conchonius has been proven. These components are classified as candidate hormones. These are not established in the classification as gastrointestinal peptide hormones or paracrine. These are termed as candidates or supposed hormones. The pancreas secretes two important hormones, insulin and glucagon. Insulin is secreted from the α-cells of the pancreatic island gland and glucagon from β-cells.
In addition to acetylcholine, evidence of the presence of vasoactive intestinal peptides and somatostatin, meta-enkephyllin, and paracrine has been found in the nerve fibers of the gastrointestinal tract (Romboult et al. 1986).
Absorption
The main technique of intestinal absorption of fish is similar to that of mammals. The material produced as a result of digestion is absorbed through diffusion and active transport. Glucose uptake through a carrier is an example of active transport. It is a mechanism by which energy is required and through which, despite the higher concentration of glucose in the cell, glucose passes through a membrane and enters the epithelial cell. In the case of facilitated diffusion, there is a carrier system that helps the compound to pass through an impermeable membrane. Similarly fructose is also absorbed through the intestinal epithelium. Facilitated diffusion does not require energy and prevents the compound from advancing against gravity.
Simple diffusion does not require any carrier or power. Fatty acids are an example of such a compound that is absorbed through the intestinal epithelium in a simple diffusion manner. Tiny particles of fatty acids and bile salts enter the epithelium. Lipid cells pass through the membrane and are released to form tiny particles called chylomicrons. This particle is surrounded by a layer of protein and dissolves in water. These chylomicrons pass through the cells, enter the bloodstream and are transported to the liver for later processing. Antibiotics affect food absorption by causing physiological changes in the gut or by the physical and chemical interactions between the drug and the food taken. Some antibiotics increase the absorption of unsaturated fatty acids in rainbow trout(Cravedi et al. 1987).
Regulatory Activities
Secretion of Digestive Juices
Most animals do not eat continuously. Fish is no exception to this rule. Usually the presence of food in the alimentary canal stimulates the secretion of digestive juices. Since fish does not have any glands like salivary glands, no digestion takes place in the mouth. The techniques and regulatory mechanisms of fish digestive juices are well known.
The presence of food in the alimentary canal immediately stimulates glandular cells and other nervous systems. This stimulus re-enters other glandular cells or organs so that they are ready to secrete enzymes. Acidic or alkaline juices do not mix with food immediately. The initiation and transport of this stimulus coordinates the secretion of digestive juices and the work of the muscles responsible for the movement of food through the alimentary canal. This process is not organized under the direct or voluntary control of the animal, but it is managed by neurological or hormonal techniques. When this secretion is controlled by a neural mechanism, it acts as a sympathetic and parasympathetic nerve arising from the vagus nerve (10th cranial nerve). The following diagrams show some of the pathways involved in the secretion of digestive juices. Some teleosts secrete histamine instead of gastrin, which regulates the physiological activity of acid secretion.
Rate of Digestion
The activity of specific enzymes and the amount of digestive juices are important for the digestive process. The following terminology is used to measure the rate of digestion:
Intestinal transit time: Difference between feeding time and 1st released faces
Intestinal emptying time: The time during taken of complete food removal
Stomach emptying time: The time between food intake and emptying stomach
These indicators are equally applicable to food digestion rates. The following methods are used to determine the digestive rate, viz:
Use of X-radiography
In this case, the fish is fed using 32P or 144Ce isotope in the food and its movement is monitored over time. It has been found that this type of food intake reduces the rate of emptying the stomach rather than the animal’s voluntary food intake.
Use of dyes
In this case, color is used in food. The stool was subsequently observed during defecation and recorded during the 1st color mixed stool visit. Hofer and Schiemer (1983) also used this method in their study of the rate at which the rate of excretion by natural populations is determined.
Direct observations
This method is used in the larval stage of the fish when the intestines of the fish and its contents are visible.
Multiple studies have been done to determine the digestive rate of different fish. Studies have shown that the digestive rate depends on different types of exogenous and endogenous regulators. They are described below:
Meal size: There has been evidence of an increase or decrease in the rate of digestion of the stomach over the size of the food. According to some researchers, large food (equivalent to the size of a fish) does not increase digestion time. According to some researchers, increasing the size of the food increases the amount of time the stomach empties, but the rate of emptying the stomach is not directly related to the increase in food size. Since enzymes and acids are required for digestion, it is not necessary to empty the stomach with food. This time depends on the temperature and is also affected by the type of food. Some of the food taken at the beginning of digestion goes to the intestines and the waste products of the stomach begin to decrease.
Temperature: Fish are said to be frozen animals and the temperature affects the rate of digestion of food. Fange and Grove (1969) found a relationship between the time and temperature when the stomach empties.
Fish size: Various experiments have shown that the digestive rate or stomach size is affected by the size of the fish during stomach emptying. Digestive rates have also been shown to decrease with increasing food size.
Type of food: The effect of diet type on emptying the stomach is clearly known. Digestive capacity not only affects the emptying of the stomach but also determines the time of weight loss of previously consumed food after ingestion. Studies have also shown that thick and hard foods (insects, larval exoskeleton, mollusk shells) reduce digestion. The rate of emptying the stomach depends on the concentration of food ingested (De Silva and Owoyemi 1983)|
Physiology of Digestion: Details about the physiology of fish digestion are not known. However, Agarwal and Singh (1962, 1964) examined the pH value of the intestinal tract and found that it was neutral or mildly acidic in Colisa fasciatus, Notopterus notopterus.The pH value of Colisa fasciatus in the stomach is about 5.6 and the pH value in the intestine is 6.7. In the case of Notopterus, the pH value in the stomach is 6.8 and the pH value in the intestines and pyloric caeeca is 6.8. According to them, most of the enzymes are secreted from the hepato-pancreas of fish.
Many researchers have found that the average pH value of 5.6 in the stomach of teleost secretes secretory digestive juices. However, the pH value of the stomach was 4.6. Pepsin and HCl are secreted from the granular cells of the gastric glands of the stomach. Moreover, the stomach of teleost contains small amounts of amylase and lipase enzymes. Stomachless fish such as Rutilus do not contain pepsin and hydrochloric acid.
In Rutilus, Gobio, Cyprinus (Al-Husseini 1949), the internal pH value is 6.12-7.2. In Fundulus, Cyprinus, Zoarces, bile is slightly acidic (pH value 5.5-6-4). A significant number of cyprinids (al-Husseini 1949) and goldfish (Carassius auratus) contain proteolytic enzymes (Sarbahi 1951). However, a clear idea about the source of this enzyme was not found. Trypsinogen is probably formed in the pancreas and erepsin and enterokinase are secreted from the intestine. The pancreatic trypsin and an intestinal enzyme of the stomachless fish are of the same nature. Amylase is produced in the pancreas of teleost.
The intestinal mucosal extract of some fish has been shown to act like amylase. Maltose and lipase have been found in the intestinal extracts of some fish. Sarbahi (1952) found lipase in liver and pancreatuc extracts. However, the source of enzyme production has not been established. There is a relationship between enzymes and animal food in the anatomy of the digestive tract. According to Al-Husseini (1949), herbivores such as Cyprinus have higher concentrations of the Hydrase enzyme and carnivorous fish Gobia have lower concentrations of this enzyme. Cyprinus, on the other hand, has a lower concentration of protease enzymes and Gobia has a higher concentration. Predatory fish have gastric glands that secrete hydrochloric acid (HCl) and pepsinogen.
Hydrochloric acid and pepsinogen combine to form active pepsin and break down protein molecules into polypeptides. In carnivorous fish such as the pike (Esox lucius), the stomach pH is measured at 2.4-3.6. Peptide enzymes have been found in the stomachs of many fish. Some Minnows (Cyprinidae) do not have gastric glands. Fish gizards do not contain any digestive enzymes. The pyloric saeca of trout contains the enzyme lactase. The pyloric saeca of carp and bluegill contain inverted sugar. Pyloric saeca, and intestinal mucosa also contain lipase. It breaks down fats into fatty acids and glycerol. Copepod-eating anchovies and herring contain 50% wax. The pyloric saeca of these fish helps in the digestion of wax.
Bile contains bilirubin and Biliverdin pigments which are produced by breaking down red blood cells and hemoglobin. Bile salt helps in hydrolysis of fats. The liver also stores fats and sugars (glycogen). Blood cells are destroyed in the liver and urea and nitrogenous substances are produced. In flatfish (Pleuronectiformes), fat is deposited in the liver. Tuna (Scombridae) and herring have a lot of fat in their muscles. In some sharks, the liver weighs about 20%. In addition to fat deposits, the liver contains vitamins A and D. Tuna contains so many of these vitamins. So if you eat these liver causes metabolic problems that cause hypervitaminosis.
The pancreas plays a role as an exocrine and endocrine gland. Insulin secretes proteases from the pyloric caeca, pancreas, and intestinal caeca that bind to the amino acids of proteins. In rainbow trout, alkaline proteases in the intestines and acidic proteases in the stomach have been found. The intestine secretes inactive zymogen enzymes. It is activated by enterokinase thus protecting the intestinal mucosa from autolysis. Different types of enzymes have been found in the intestinal and pancreatic juices of fish to digest special carbohydrates. Herbivorous fish(Oreochromis) contains activated amylase activity across the gastro-intestinal tract, but the pancreas of carnivorous fish is the only source of amylase.
Some fish, especially Menhaden (Brevoortia), Silver Side (Menidio) and Silver Perch (Bairdiella) contain endemic bacteria. These bacteria contain cellulose enzymes which break down the cellulose of plant material taken by fish into small material. Examination of feces of herbivorous fish such as grass carp (Ctenopharyngodon idellus) has shown that the cell wall of plants is not broken by teeth. Green particles can be seen in their stools. In the case of sharks and races, the sight and smell of food do not secrete gastric glands, and the opposite condition is seen in mammals.
Mechanism of Feeding
Most teleosts receive oral food by sucking and expanding the oral cavity and gill pouches (Alexander 1967). The mouth cavity and gill pouch are very important for sucking the surroung water pressure. It is known that the suction of food is completed under the pressure of 50-105 cm of water and the 1-9 cm of water that comes with the food is expelled through the operculum. In the case of Black Bullhead (Ictalurus), a negative pressure of 80 cm at 180 C has been recorded. It has also been shown that this strong negative pressure maintains the highest isometric tension by engaging all types of muscles in creating stress (Alexander 1970). Osse (1969) made an electromyograph on food intake of Perca and confirmed the sequence of muscle action by examining the similarity and structure of this process with respiration.
Stimuli for Feeding
The feeding strategy of fish is quite complex. There are several stimuli for food intake. There are two types of stimuli for food, namely:
(1) different factors such as season, length of day, light penetration, last meal time, temperature, etc., and
(2) different sensations such as smell, taste, vision, sensations received through the laterral line organs create food stimulation.
Both factors collectively control the feeding process of fish (Lagler et al.1977). In nocturnal fish such as bullheads (Ictalurus), smell and taste play an important role in food intake.
On the other hand, diurnal fish such as pike (Esox) and other predatory fish are more efficient in the daytime, so vision plays a major role in food intake. In non-tropical regions, seasons affect water temperature, but some fish, such as salmon (Salmo, Onchorhynchus) and lamprey (Petromyzonidae), stop eating during the breeding season. Among other fish, the Southeast Asian swamp eel (Synbranchidae) does not eat for several weeks while spending time in damp holes in the mud for summer sleep. At this time they eat the stored fat. Fish in the temperate zone actively feed in the spring.
In dogfish (Squalus), Man eating Shark (Carcharodon) etc. chemical attraction helps in food absorption. Murays eels (Gymnthorax) search and select food by smell. Catfish (Ictaluridae) and goatfish (Mullidae) mainly eat by taste, but touch also helps in this. Food size, color, movement, etc. affect food intake. The penetration of light into the water also plays an important role in food intake. Yellow perch (Perca flavescens) and some other fish eat all day. Appetite controls by the hypothalamus of the brain. Food intake depends on metabolism with a decrease in body growth resulting in maximum growth in spring, rapid growth in summer, and reduced growth in autumn and winter.
Groot’s (1971) study found that food intake is involved in sight, chemical, and mechanical sensory organs in the fish of the family Pleuronectidae, Soleidae and Bothidae (Pleuronectiformes).
Fish of the family Soleidae intake food as polychaetes mollusca etc. They eat at night and usually search for food through the sense of smell. But they can also search for food through sight. Barbell helps fish to search for insects, larvae, etc. from the bottom of the pond.
Table: The role of eye and olfactory organs in food intake
|
Family |
Type of food |
Methods of food search |
Structure of intestine |
Structure of gill rakers |
Olfactory lobe( (After Evans 1937))
|
Optic lobe (After Evans 1937) |
|---|---|---|---|---|---|---|
|
Bothidae |
Different fish |
Sight |
With simple loops |
With strong teeth |
Small |
Large |
|
Pleuronectidae |
Crustacea |
Mainly sight but smell also plays a role |
With complex loops |
With fewer teeth |
Medium |
Large |
|
Soleidae |
Polycjaetes and mollusca |
Smell, but sight plays a role |
With more complex loops |
Few or toothless |
Large |
Small |
You might also read: Digestive System of Fishes